 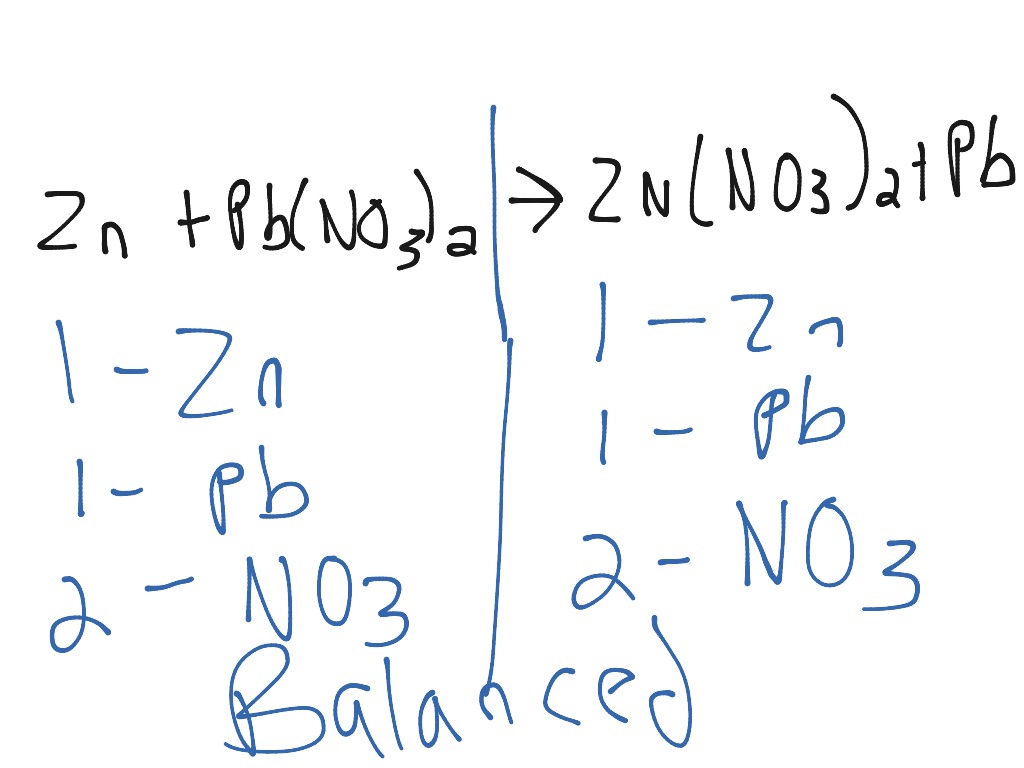
When heated to about 320 C (608 F), silver nitrate loses oxygen and forms silver nitrite. It is moderately soluble in methyl and ethyl alcohols and to a lesser extent in various other organic solvents. In this reaction, the NO 3 - ions remain in the solution and do not react - they are the spectator ions in this reaction. The solubility at 20 C (68 F) is 222 grams per 100 grams of water. For example, copper reacts with silver nitrate solution to produce silver and copper(II) nitrate solution:ĢAgNO 3 (aq) + Cu(s) → 2Ag(s) + Cu(NO 3 ) 2 (aq) Silver-ammonia solution This stock solution is prepared from silver nitrate, sodium hydroxide and ammonium hydroxide (5 M Aldrich): here, again, the use of titrated solutions is strongly. Ī common type of displacement reaction takes place when a reactive metal reacts with the salt of a less reactive metal.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
